Due to the increasing use of disposable paper packaging (W-Pack) in medical centers and the importance of sealing, it is necessary to use a sealer device to seal this type of packaging paper. The sealer device must be evaluated daily. When we put an instrument in disposable papers and sterilize it in an autoclave, we must make sure that after removing the package until it is used in the surgical room, the ambient air does not enter the package and does not take it out of the sterile state. Therefore, the correct operation of the sealing and packaging machine should be evaluated. The Ink Sealer Device Test (RRS 14-81210) is a standard and ideal method for diagnosing porosity and correct sealing in paper and sterile medical packages (V-pack).
Instruction for use
- Turn on the sealer device and allow it to reach to its appropriate operating temperature
- Separate about 10 cm from the roll-pack for sterile packaging.
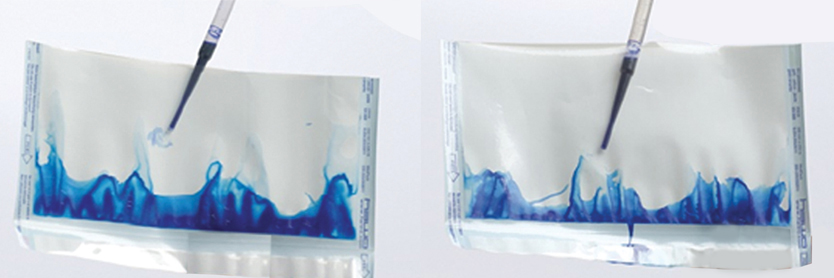
- Using a dropper, remove at least 2 ml of the detection ink and pour it through the open part. Then rub the package with your hand to spread the ink all over the package.
- After 20 seconds, check the closed seams to make sure those are sealed.
- Ink leaks in the seams will be easily visible.
Result: If the ink does not penetrate into the sealed part, it indicates the sealing is healthy.
Note
The quality of the seal stages should be validated using ink and paper tests and then archived. Having a regular monitoring program helps to quickly detect any changes or problems in the packaging process.
Advantages
- Confirm that the packages are sterile during storage
- Ensure the correct sealing of the sealer device
- Quick and easy to use and low costs
Download Catalog
Please Check out file at the following link
Hemo test (Indicator of Monitoring the Washing Process) (RRS 14-42110)
Protein Test (Indicator of Monitoring the Washing Process) (RRS 14-41110)
Automatic Rotary Sealer (R900) (RRS 14-3410)
Marker with indicator ink for steam sterilization process RRS



